New 3-HRP conjugate
Advancing Biochemical Analysis: Introducing the Advanced eQuant-HRP
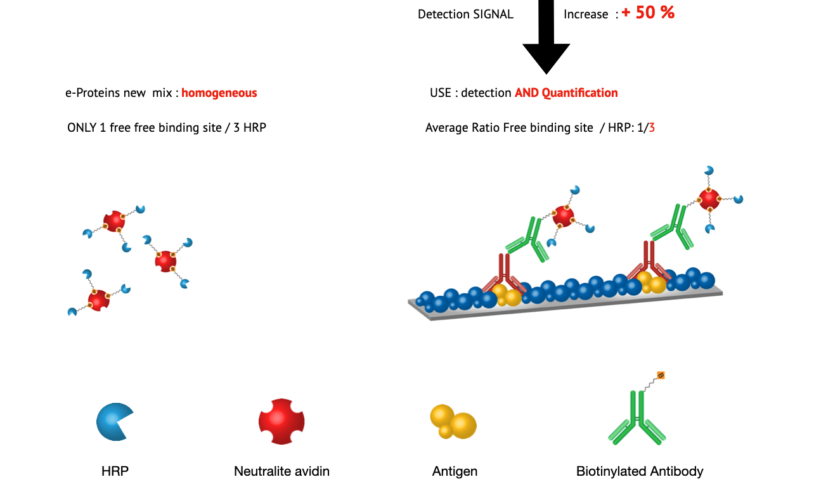
Optimized Ratio of HRP to Biotin-Binding Protein
conjugate over current market offerings. Firstly, it uniquely enables direct quantification of biotinylated elements. Secondly, we have engineered eQuant-HRP to achieve an optimal balance between Horseradish Peroxidase (HRP) and biotin-binding protein. Thirdly, this carefully calibrated balance significantly enhances specificity in various assays, leading to more accurate and reliable detection.
Enhanced Efficiency and Direct Quantification
Our design of eQuant-HRP enables more efficient binding to biotinylated targets. This conjugate’s ability for direct quantification ensures straightforward and less time-consuming detection and measurement of biotinylated elements, especially beneficial in high-throughput settings where speed and accuracy are critical.
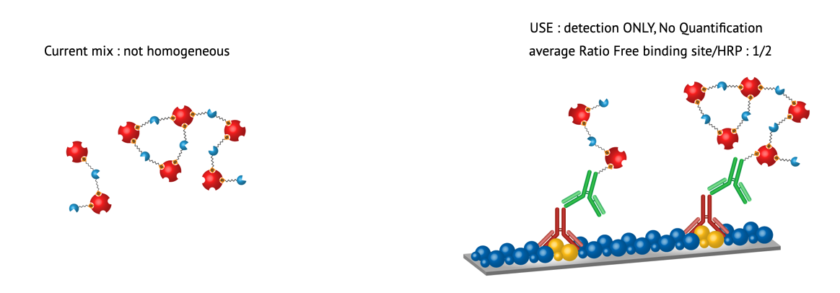
Significant Reduction in Non-Specific Binding
The improved HRP to biotin-binding protein ratio in eQuant-HRP significantly reduces non-specific binding. This reduction not only increases the accuracy of assay results but also minimizes potential interference, providing clearer and more definitive outcomes.
Conclusion: Advancements in Biotin-Binding Protein Technology
In summary, the introduction of eQuant-HRP marks a significant advancement in biotin-binding protein technology. Its optimized ratio of HRP to biotin-binding protein, leading to decreased non-specific binding, and its capability for direct quantification, make it a superior choice for biochemistry professionals.
prix
On request
specification
not available on the net
sourcing
High-Quality Enzyme Sourcing and Development
CLIA Application
HRP finds use in ChemiLuminescent ImmunoAssays (CLIA). This technology merges chemiluminescence’s sensitivity with immunoreaction specificity. HRP transforms a substrate into a chemiluminescent signal, emitting photons in the visible or near-visible range. The light produced directly correlates with the target analyte’s quantity in the sample. The homogeneity of 3HRP enables direct quantification.
CLIA holds a significant advantage over the Elisa Method. It is far more sensitive due to signal amplification and multiplication. Moreover, it operates without the need for incubation time or stopping reagents.
